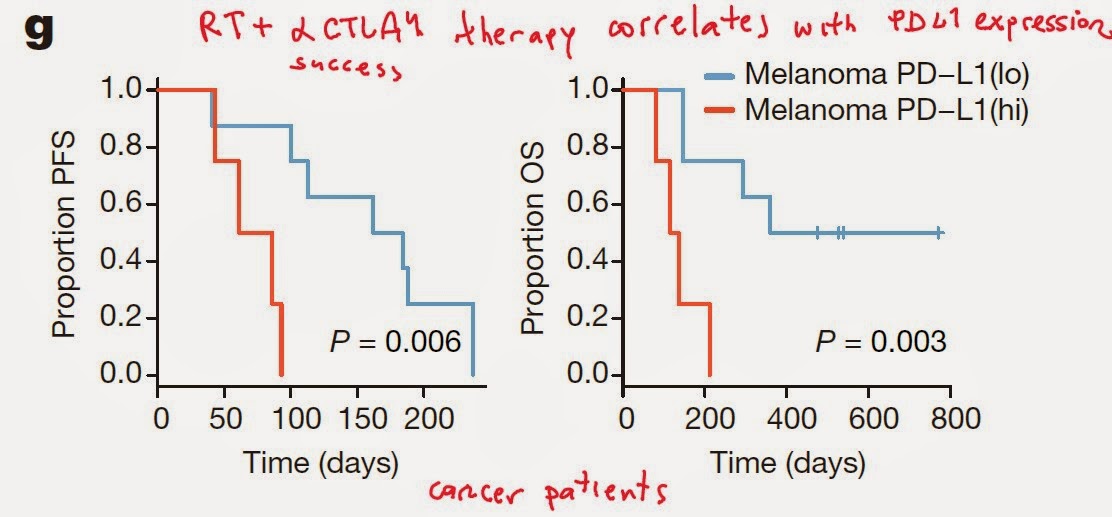
Introduction of checkpoint inhibitors, α-CTLA4 and α-PD1/PD-L1 humanized antibodies, injected new hope in cancer immunotherapy. Still, these clinical advances are at early stage and the significant benefits are achieved in small proportion of patients (~ 20%, for solid tumor patients).
Why is that? According to new study in journal Nature, it appears that success of tumor immunotherapy requires simultaneous deployment of parallel checkpoint inhibitors, in addition of locally focused radiation therapy.
The concept is similar to treatment of antibiotic resistant pathogenic microorganisms. Single mechanism of action usually is not sufficient for successful treatment since microorganisms are quick to adapt and develop resistance to it.
Tumors, too, frequently behave as the separate macro-organisms within the body. When coming under immune pressure exerted by α-CTLA4 antibody, tumors will try to escape immune surveillance by up-regulating parallel inhibitory pathways such as PD1/PD-L1 system.
Identification and simultaneous targeting of such inhibitory and/or escape strategies will finally provide clearly measurable clinical benefits in majority of tumor patients.
David Usharauli

No comments:
Post a Comment